Molecular Therapy of Cancer
A new agent that treats more than just breast cancer has been permitted for human testing. The information was confirmed by the
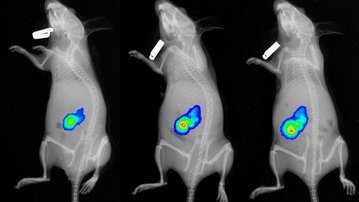
At the beginning of last year, researchers from the Institute of Biotechnology of the Czech Academy of Sciences at the BIOCEV Center published great news. In collaboration with the Czech company Smart Brain s.r.o. (the KKCG Group), they developed MitoTam, an agent that managed to entirely eliminate cancerous cells of one of the most aggressive types of breast cancer. The preparation has successfully passed laboratory tests and clinical trials. Now, the State Institute for Drug Control granted permission for conducting clinical trials, which will take place at the General University Hospital in Prague under oncologist Prof. Luboš Petruželka.
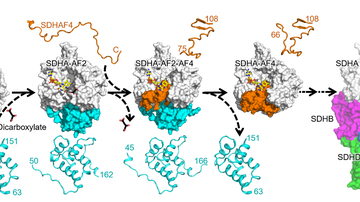
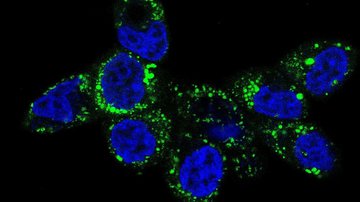
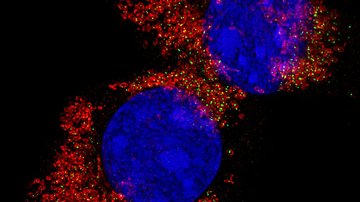
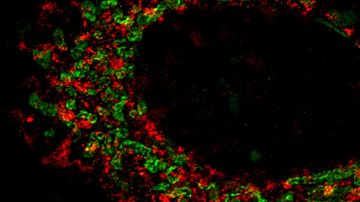
Mitochondria-focused MitoTam significantly reduces the occurrence of breast cancer and is also effective against other types of cancer. The research conducted by Prof. Jiří Neužil and his team from the Institute of Biotechnology was an essential part in the development of this new anti-cancer agent. Together they brought about new biological knowledge important for understanding mitochondria as the “power plants” necessary for the work and life of both healthy and cancerous cells. Their comprehensive research helped identify weak spots in cancer cells and suggested a method of how to attack these weak spots1. This was followed by demanding chemical analyses carried out by Jan Štursa and Lukáš Werner, who designed and synthesized all tested agents. One of the proposed structures, MitoTam, has proven to be a potential medication able to disable the function of mitochondria and initiate a programmed death of cancer cells.
“Every year, tens of thousands of patients are diagnosed with cancer in the Czech Republic. We are proud that we can be part of something so helpful and unique,” says K. Komárek from Smart Brain s.r.o.
The Service Technology Laboratory at the Institute of Biotechnology, engaged in the “Pre-clinical Testing of Potential Medications” and the Centre of Preclinical Testing under the Academy of Sciences’ Strategy 21 has played an important role int the pre-clinical test. The Service Technology Laboratory also helped to synthesize MitoTam to a medicinal quality and helped prepare the documentation for a clinical trial. For the transition of MitoTam towards practical application, the involvement of Karel Komárek was crucial. Komárek is the founder of Smart Brain s.r.o., which has supported interesting research projects all over the Czech Republic for 8 years.
“Without financial aid and enormous energy and persistence when coordinating the project, the smooth transition from the laboratory to a patent application to a clinical trial would not be possible. Throughout the project, the involvement of KKCG guaranteeing promising perspectives for MitoTam was crucial, “ add the Director of the Institute of Biotechnology, Jana Pěknicová.
1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5206771/