Imaging Methods
How fast do molecules move?
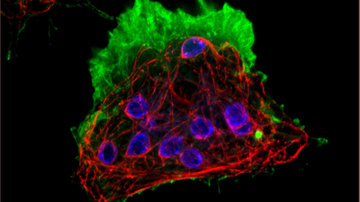
Microscopy is often associated with static images providing details of the structure of a sample. But living nature is very dynamic and merely a structure without any dynamics reveals only part of the secret. We know where is what located, but we have no idea what exactly it does there. Is it a tightly bound structural molecule or a freely diffusing protein? Does it behave indifferently with its surroundings or does it actively interact with it? What possibilities does microscopy provide us for capturing motion and thus for revealing the real function of molecules?
Everyone will probably think of the possibility of collecting a time sequence of images, i.e. shooting a video. Modern sensitive and fast cameras can capture over 100 images per second, and confocal microscopes are able to obtain multiple images per second. This speed makes it possible to accurately characterize the changes in the structure of cells, the reconstruction of their internal arrangement. However, how can we capture and characterize the motion and displacement of molecules that may not necessarily be directly related to specific structures?
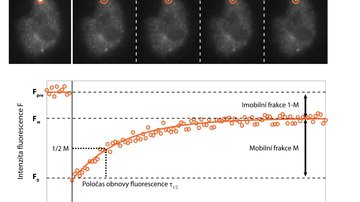
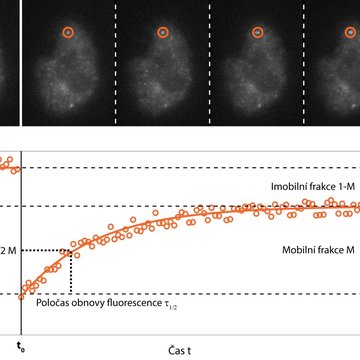
One of the possibilities is to supplement fast imaging with targeted photo-manipulation of a sample. The Fluorescence Recovery After Photobleaching (FRAP) method specifically destroys fluorescent molecules in a selected portion of a sample using strong irradiation, usually with a laser, and monitors how quickly and to what extent the signal is recovered in a subsequent rapid scan. This provides information on the speed and mode of movement of the monitored molecules, their interaction with the environment, and possibly the speed of their cell synthesis.
A complementary technique is selective photoactivation approach, which has recently become quite popular. Using genetic manipulations special fluorescent proteins that do not fluoresce in their native state have been developed. To activate them, it is necessary to irradiate them with a specific type of light, which will only switch them to a fluorescent state. By irradiating a selected part of the sample, it is thus possible to photo activate only a small portion of the molecules’ population and subsequently monitor whether the molecules remain in place or move to other parts of the cell. Compared to photo-bleaching, it is sufficient to use orders of magnitude lower intensities with a lower risk of phototoxicity for the observed living cells.
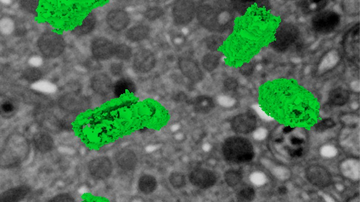
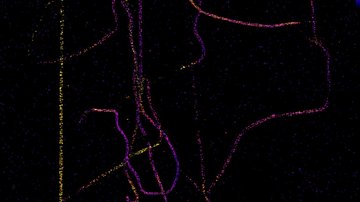
Both of the above discussed methods take advantage of the perturbation of the system out of equilibrium and monitor how the system copes with the change. Is it possible to monitor the dynamics of molecules even without artificial imbalance? Under certain conditions, yes. If the sample can be labelled with an extremely low concentration (of the order of picomolar) of fluorescently labelled molecules of interest that do not move too fast, and the fluorescent label is sufficiently bright and stable, the movement of individual molecules can be monitored using an extra-sensitive and fast camera. The method called Single Particle Tracking (SPT) is applied mainly to membrane bound molecules and by analysing the trajectories of individual molecules, it is possible to measure not only the speed of movement or diffusion, but also to detect possible specific interactions.
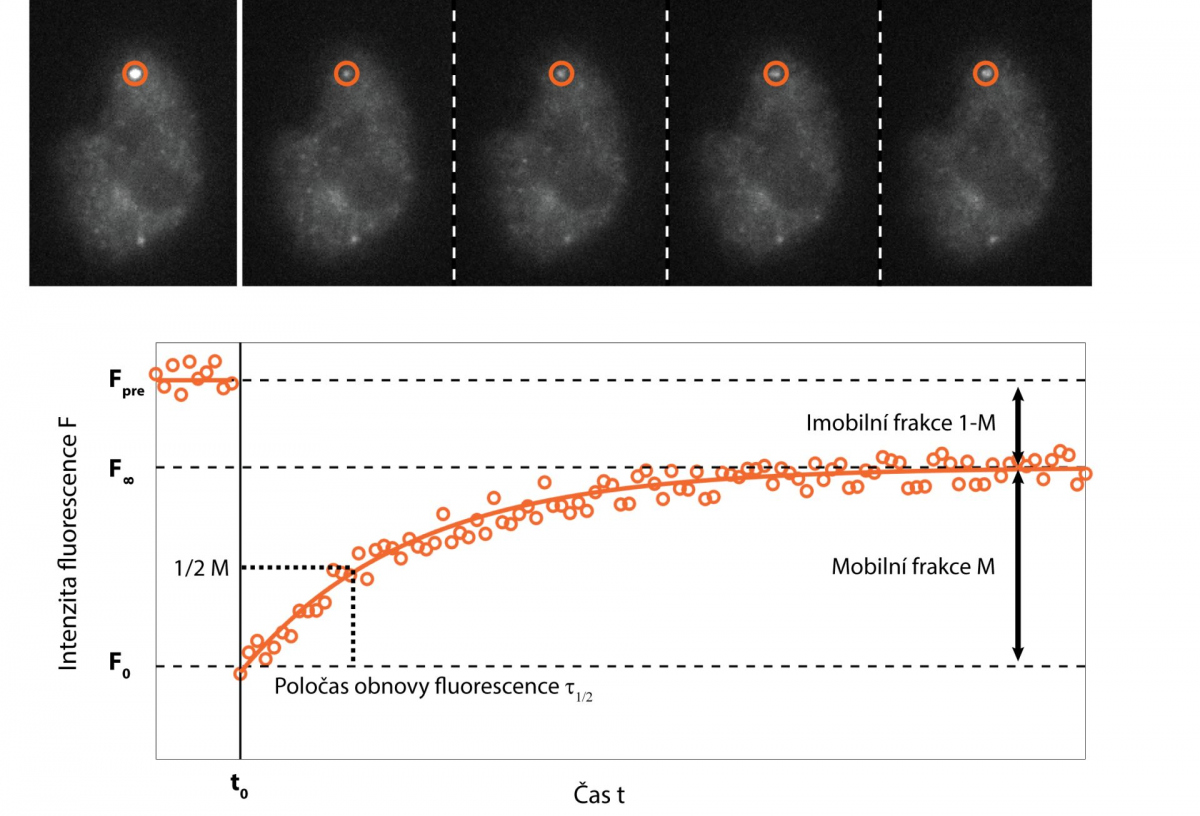
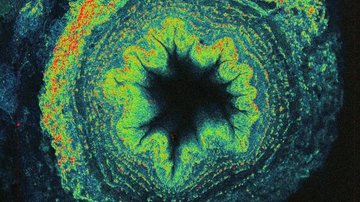
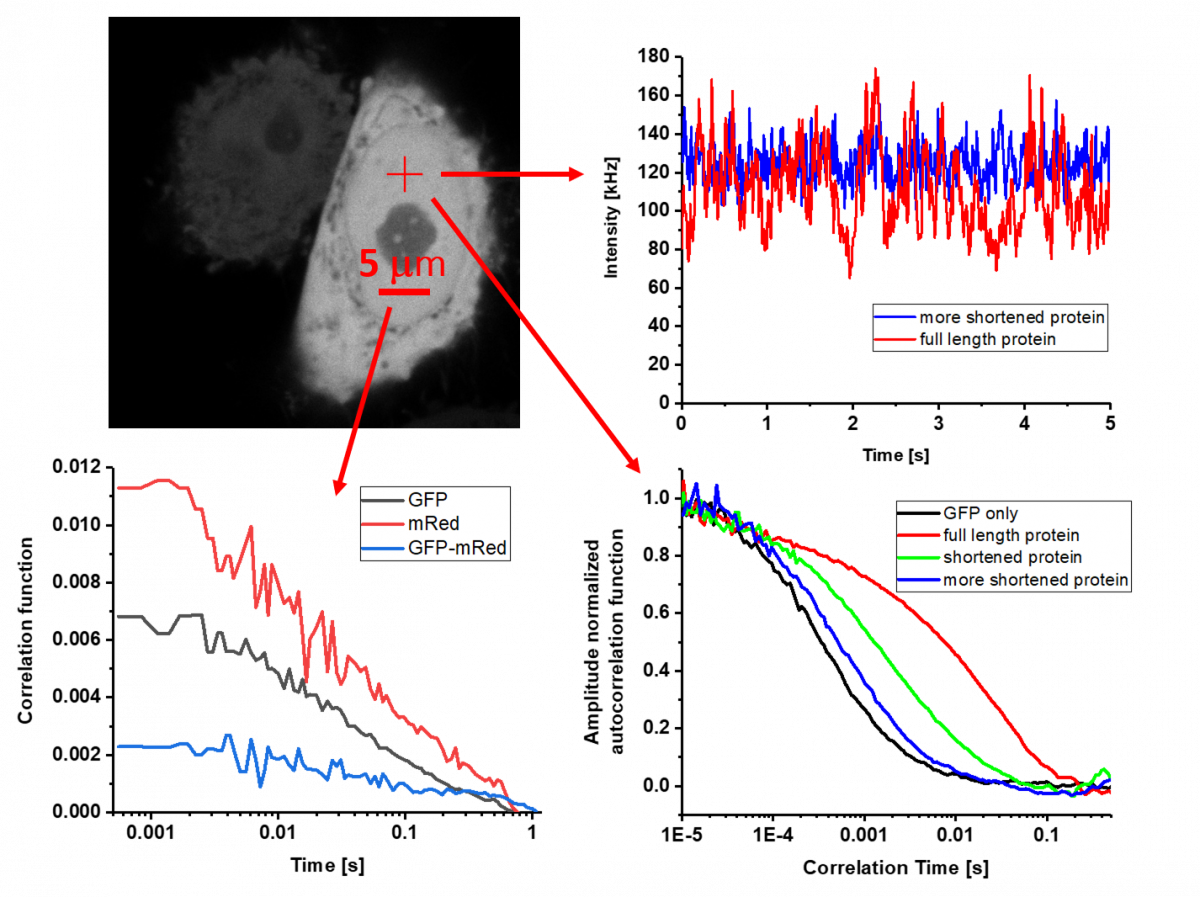
If the studied sample does not meet the somewhat strict criteria for SPT, it is possible to use spatio-temporal correlation techniques. The name of the method sounds complicated, but the principle is relatively simple. The method monitors whether the measured fluorescence intensity at one point and / or time of the sample is somehow related (i.e. correlated) to the fluorescence intensity at another point and / or time. When signals are related, it usually means that they are affected by the motion of the same molecules.
Although at first glance it often seems that we analyse only noise, from this "noise" we obtain information about the concentration of molecules, the speed and mode of their movement, as well as about possible interactions or conformational changes. The condition is that the concentration of fluorescent molecules is so low that the change caused by one molecule is distinguishable from real noise. The most common implementation is point fluorescence correlation and cross-correlation spectroscopy (point FCS and FCCS), which uses confocal microscopes with very sensitive detectors capable of accurately registering the detection of a single photon.
The extremely small femtoliter detection volumes generated by the best lenses on the market make it possible to analyse up to one hundred nanomolar concentrations, which are relevant to the natural cellular environment. The advantage of point FCS is that it can cover processes on a time scale from nanoseconds to units of seconds. If we observe slower movements and we would like to know the spatial distribution of the obtained parameters, it is possible to successfully apply line-scanning FCS and Imaging FCS, as well as other derived methods. Czech-BioImaging offers some unique methods in this area, developed by experts in service laboratories, which are not available anywhere else in the world.
Author: Aleš Benda, Head of Imaging Methods Core Facility
The mentioned procedures, techniques and equipment are operated by the following workplaces: The Institute of Physiology, The Academy of Sciences of the Czech Republic - Bioimaging Facility, Masaryk University (CEITEC) - Cell Imaging, Charles University (BIOCEV) - Imaging Methods, Palacky University in Olomouc - The Department of Molecular and Translational Medicine - The Microscopic Imaging Facility, The Institute of Experimental Botany, The Academy of Sciences of the Czech Republic - The Imaging Facility, The Institute of Molecular Genetics, The Academy of Sciences of the Czech Republic - Light Microscopy, Brno University of Technology (CEITEC) - Experimental Biophotonics.