Structural biology of signaling proteins
Ph.D. positions available for motivated students
PhD project: Molecular mechanisms of 14-3-3 proteins in signaling complexes
Laboratory website: http://www.biomed.cas.cz/d312/
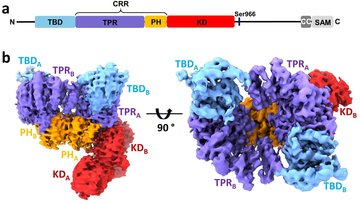
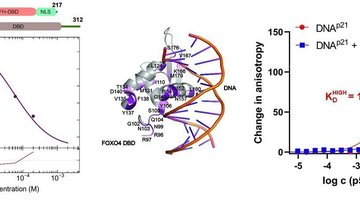
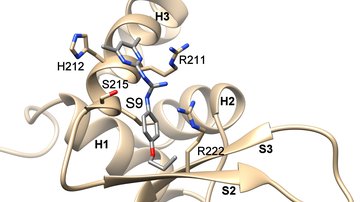
14-3-3 proteins have the ability of binding the functionally different signal proteins, including kinases, phosphatases and transmembrane receptors by changing their function. Through the functional modulation of a wide range of binding partners, 14-3-3 proteins are involved in many important physiological and pathophysiological processes, including cell cycle regulation, metabolism control, apoptosis, and control of gene transcription. The scope of this PhD project lies in the field of structural biology/biophysical chemistry.
The main focus of this project will be on elucidation of mechanisms by which the 14-3-3 proteins regulate selected binding partners using a wide range of biophysical approaches. The work plan of the project contains preparation of recombinant proteins, biochemical and biophysical characterization of protein-protein interactions, and structural characterization of selected protein complexes, using protein crystallography, small angle x-ray scattering (SAXS) and fluorescence spectroscopy. The research will be performed in close cooperation with the Faculty of Science, Charles University.
References:
- Pohl P, Joshi R, Petrvalska O, Obsil T* and Obsilova V* (2021) 14-3-3-protein regulates Nedd4-2 by modulating interactions between HECT and WW domains. Commun. Biol. 4(1):899. https://www.nature.com/articles/s42003-021-02419-0
- Lentini Santo, D., Petrvalska O., Obsilova V., Ottmann C. and Obsil T. (2020) Stabilization of protein-protein interactions between CaMKK2 and 14-3-3 by fusicoccins. ACS Chem Biol. 15(11):3060-3071. https://pubmed.ncbi.nlm.nih.gov/33146997/
- Hagenbuchner, J, Obsilova, V, Obexer, P, Obsil, T*, Ausserlechner, MJ* (2021). Discovery of Small Compounds that Target FOXO Transcription Factors and Modulate their Transcriptional Activity and Physiological Function. Book chapter in “New Innovations in Chemistry and Biochemistry” Vol. 4, pp 41-54. https://doi.org/10.9734/bpi/nicb/v4/14349D
- Kalabova D+, Filandr F+, Alblova M, Petrvalska O, Horvath M, Man P, Obsil T*, Obsilova V* (2020) 14-3-3 protein binding blocks the dimerization interface of caspase-2. FEBS J. 287:3494-3510. https://pubmed.ncbi.nlm.nih.gov/31961068/
- Hagenbuchner, J+, Obsilova, V+, Kaserer T+, Kaiser N, Rass B, Psenakova K, Docekal V, Alblova M, Kohoutova K, Schuster D, Aneichyk T, Vesely J, Obexer P, Obsil T*, Ausserlechner MJ* (2019) Modulating FOXO3 transcriptional activity by small, DBD-binding molecules. Elife 8, pii: e48876. https://pubmed.ncbi.nlm.nih.gov/31789593/
- Smidova A+, Alblova M+, Kalabova D+, Psenakova K, Rosulek M, Herman P, Obsil T*, Obsilova V* (2018) 14-3-3 protein masks the nuclear localization sequence of caspase-2. FEBS J. 285:4196-4213. https://pubmed.ncbi.nlm.nih.gov/30281929/
- Alblova M, Smidova A, Docekal V, Vesely J, Herman P, Obsilova V*, Obsil T* (2017) Molecular basis of the 14-3-3 protein-dependent activation of yeast neutral trehalase Nth1. Proc Natl Acad Sci U S A. 114:E9811-E9820. https://pubmed.ncbi.nlm.nih.gov/29087344/
Project Supervisors (email): Veronika Obsilova, Ph.D. (veronika.obsilova@fgu.cas.cz), prof. Tomas Obsil, Ph.D. (obsil@natur.cuni.cz)
Candidate´s profile (requirements): Successful candidate should enjoy working in the field of structural biology/biophysics/physical chemistry, be curious and has the courage to try out new things. Required is M.Sc. degree or those expecting to obtain their degree this year (fields: Chemistry, Biochemistry, Molecular Biology or similar). The knowledge of molecular biology, protein expression and purification is a strong advantage. Good knowledge of English is required. We offer: friendly, motivating environment, part-time/full-time contract for 3 years with the possibility of renewal, 5 weeks of vacation, subsidized lunch.