About us
Quantitative live-imaging, cytoskeletal dynamics, nematode genetics, biophysics, evolutionary development (evo-devo)
The shape of an animal arises in a species-specific, step-wise fashion during embryonic development. During this sequence of events, collectively referred to as ‘embryo morphogenesis’, the embryo constantly remodels its shape. Our lab is interested in the force-generating mechanisms that drive these shape changes.
We aim at addressing the following questions:
- How do molecular-scale forces arise in developing embryos?
- How do molecular-scale forces drive embryonic shape changes?
- How do these force-generating mechanisms change on evolutionary time scales?
Our research operates at the intersection between cell biology, evolutionary developmental biology (evo-devo) and biophysics, and employs quantitative methodology from all these disciplines.
The forces that drive embryo morphogenesis are generated by the cytoskeleton of embryonic cells and mainly arise in the actomyosin cortex: a dense two-dimensional network of cross-linked actin polymers residing directly underneath the plasma membrane (see figure 1). By pulling and twisting, actin polymers, myosin motors and numerous accessory proteins can generate active forces within the cortical layer. Tight spatiotemporal regulation of these molecular-scale forces in developing embryos ultimately results in cellular-scale shape changes required for embryo morphogenesis.
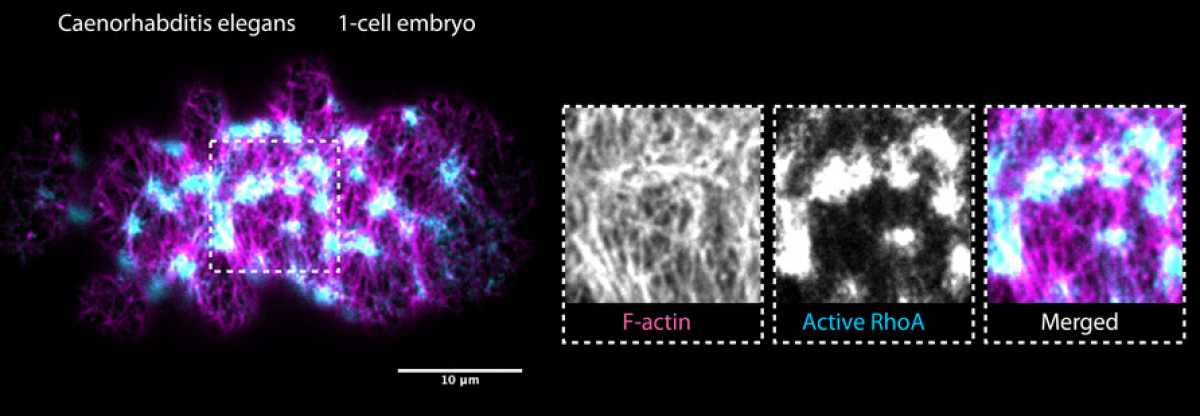
We use Caenorhabditis elegans and related nematode species to study how molecular forces generated within the actomyosin layer give rise to morphogenetic shape changes in early embryos. To this end, we combine the strength of C. elegans genetics with time-lapse imaging of early embryos (both at high-resolution and at super-resolution), quantitative image analysis and biophysical modelling.